
by Andrew Turner | Oct 11, 2021 | Clinical Trials, Good Clinical Practice
What are monitoring and centralised monitoring and how do they differ from remote monitoring? Monitoring of a clinical trial is an essential requirement of GCP and ISO 14155 and is the responsibility of the trial sponsor. The...

by Andrew Turner | Oct 11, 2021 | Clinical Trials, Good Clinical Practice
What is a Trial Master File? During a clinical trial, a multitude of essential documents are collected to manage the study and verify whether the trial has been conducted in line with the applicable regulatory requirements and Good Clinical Practice (GCP)...
by Andrew Turner | Jun 19, 2020 | Clinical Trials, Good Clinical Practice
Randomised controlled trials (RCTs) are considered the gold standard for studying the safety and efficacy of new interventions and are generally required by regulatory bodies for approval of new therapies. RCTs reduce bias by controlling both known and unknown...
by Andrew Turner | Apr 21, 2020 | Clients, Clinical Trials
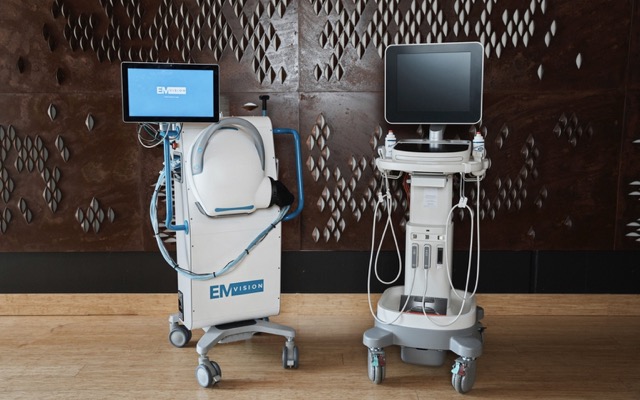
EMVision Medical Devices (ASX: EMV) has released what it claims is “promising” first stroke patient images from a pilot clinical trial that aims to improve the understanding of stroke on electromagnetic scattering effects in the brain. According to the portable...

by Andrew Turner | Mar 13, 2019 | Clinical Trials, Good Clinical Practice
Australia is already an attractive location to host clinical trials with an efficient ethics and regulatory framework, diverse participant recruitment pool and high-quality research facilities just a few of the drawcards. Clinical trials contribute over $1.1bn...

by Andrew Turner | Mar 21, 2018 | Clinical Trials
Services are often outsourced to third party vendors for certain clinical trial activities, particularly where a clinical trial activity must be standardised. Such vendor services can include a centralised review of imaging data, central pathology or laboratory...








