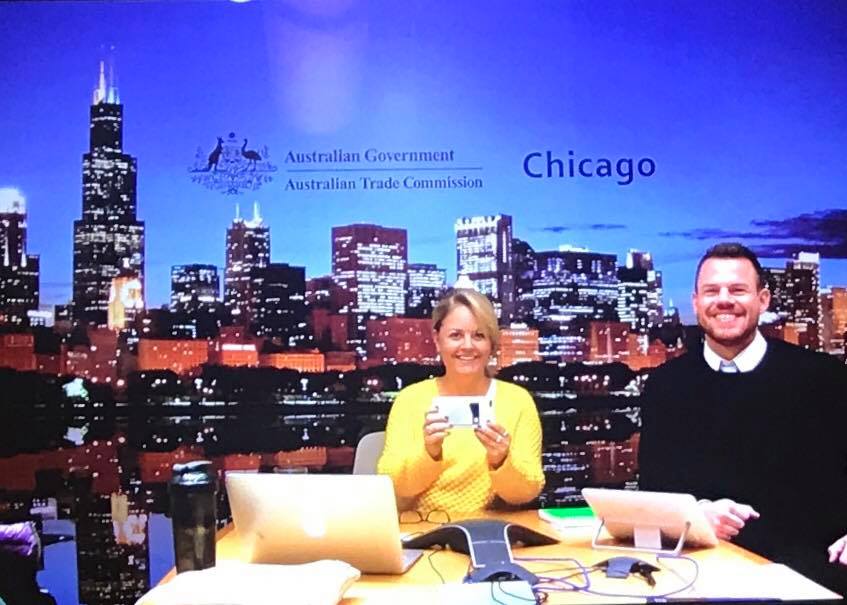
by Andrew Turner | Feb 2, 2018 | Clinical Trials
AusTrade’s latest Clinical Trials Capability Report has some interesting facts n figures Between 2012 and 2015, phase I activity has grown by 17.2% in Australia, compared to 1.8% globally Between 2012 and 2015, phase I activity has grown by 17.2% in Australia,...

by Andrew Turner | Dec 11, 2017 | Clients, Clinical Trials
We are please to report that AbilityMate has won the 2017 National Disability Excellence in Innovation Award “We’re living in an era of design where human invention, empowered by technology could reshape the lives of people with disabilities for the better.” Melissa...

by Andrew Turner | May 8, 2017 | Clients, Clinical Trials
Mobius Medical, like many other CROs all over the world, is privileged to be involved in the development of a myriad of novel innovations, and life changing medical devices, diagnostic tools and medicines. We are in the business of providing clinical trials...

by Andrew Turner | May 2, 2017 | Clinical Trials, Electronic Data Capture, Good Clinical Practice
Since the GCP (E6 R1) was first released in 1996, clinical trials have evolved substantially in complexity, technological capabilities, and costs. ICH E6(R1) has been amended to encourage implementation of improved and more efficient approaches to clinical trial...
by Andrew Turner | Mar 14, 2017 | Clinical Trials, Good Clinical Practice
For many years, Australia has been the one of the most attractive countries to perform clinical investigations for medical devices, pharmaceuticals and in-vitro diagnostics. The Clinical Notification Scheme (CTN) is a straight-forward and fast approval process from...
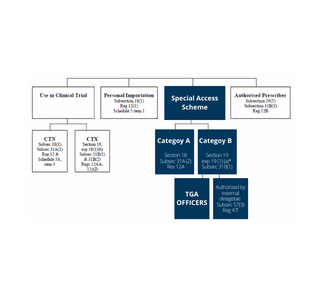
by Andrew Turner | Jun 16, 2016 | Clinical Trials
Principal investigators are often asked by their patients if they can still access the investigational product (IP) after they have completed the clinical trial. In some medical device trials, using an implantable product for example, typically the product is provided...








